by Xander F. van Kooten, Federico Paratore (Israel Institute of Technology and IBM Research – Zurich), Moran Bercovici (Israel Institute of Technology) and Govind V. Kaigala IBM Research – Zurich)
Highly sensitive and specific biochemical assays can provide accurate information about the physiological state of an individual. Leveraging microtechnology, we are developing miniaturised analytical tools for precise and fast biomolecular analysis. This work is being performed within the scope of the EU-funded project ‘Virtual Vials’ between IBM Research – Zurich and Technion – Israel Institute of Technology in Haifa.
In vitro diagnostics is a rapidly growing field and market that encompasses clinical laboratory tests and point-of-care devices, as well as basic research aimed at understanding the origin of diseases and developing improved testing methods and treatments. The global market for in vitro diagnostics is projected to grow to $75 billion by 2020. The development of novel tools that enable fast and sensitive analysis of biological samples with high throughput are essential for enabling discoveries and providing better care for patients.
In the past two decades, in vitro studies have shifted rapidly from classical tools to microfluidic devices (often also referred to as “lab-on-a-chip”). Microfluidic devices enable new capabilities and higher efficiencies in the analysis and control of biological liquid samples as many physical phenomena scale favourably as size is reduced. Effects such as diffusion and surface tension dominate the behaviour of fluids and solutes at micrometre length scales, while inertia and body forces such as gravity are negligible.
One of the key applications of in vitro diagnostics is the detection of biochemical species, such as proteins or nucleic acids that indicate a physiological condition or the presence of a disease. In many cases, the relevant analytes are present at such low concentrations that they cannot be detected using conventional methods. To overcome this, we make use of a specific electrokinetic separation and focusing technique called isotachophoresis (ITP). Isotachophoresis is a form of electrophoresis that is almost a century old, but has attracted renewed interest in the past decade, largely thanks to the strong growth of microfluidics research.
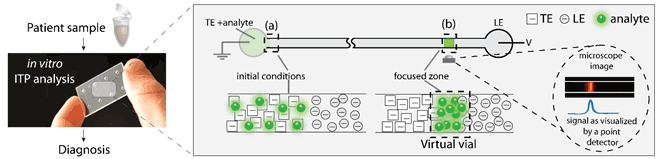
Figure 1: Schematic of ITP as a tool for in vitro diagnostics. Biochemical analytes from patient samples can be focused using ITP to increase their concentration locally. ITP uses a discontinuous buffer system to focus analytes at the interface between two electrolytes under an applied voltage V. This facilitates their detection and can lead to a faster and more sensitive diagnosis.
ITP uses a discontinuous buffer system to separate and concentrate target analytes based on differences in their electrophoretic mobility. As illustrated in Figure 1, the discontinuous buffer system consists of a leading (LE) and a terminating (TE) electrolyte, which respectively have a higher and a lower electrophoretic mobility than the analyte(s) of interest. When an electric field is applied, the analytes are focused at the continuously moving interface between the LE and TE. In this way, a “virtual vial” of several hundred picolitres is created, in which the concentration of analytes is enhanced by many orders of magnitude.
We focus on sensitive and rapid detection of proteins, which is key for early diagnosis of a large number of medical conditions. Protein analysis is commonly performed using surface immunoassays, which use antibodies immobilised on a surface to capture target proteins from a liquid sample. The number of captured proteins is converted into a readable signal by a variety of methods, such as electrochemical reactions, fluorescence or surface plasmon resonance. However, the performance of all these detection methods is limited by the kinetics of the protein-antibody reaction because the reaction time is inversely proportional to the concentration of proteins.
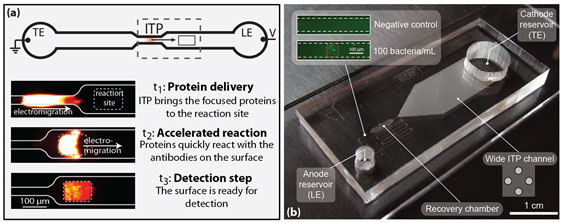
Figure 2: Two applications of ITP for in-vitro diagnostics we pursue. (a) In an ITP-based immunoassay, proteins are focused and delivered to a reaction site, where the enhanced concentration drives an accelerated reaction. (b) A device for ITP focusing from large sample volumes. The inset shows bacteria focused from an initial concentration of 100 bacteria/mL.
We use ITP to accelerate the surface reaction more than 1000-fold, so that a reaction that would require more than 20 hours under simple mixing can now be completed in under one minute. Figure 2a shows fluorescence images of the ITP-based immunoassay: At (t1) a fluorescent protein accumulates at the moving ITP interface; at (t2) the focused proteins reach a 100 µm long reaction site, where the accelerated reaction takes place, and at (t3) the proteins are carried away by ITP, leaving the reacted surface ready for detection. Combined with standard detection molecules available on the market, the technique can increase the sensitivity of existing immunoassays 1,300 times.
While ITP has proved to be a useful technique for accelerating reaction kinetics, the gains of this approach are ultimately limited by the volume of sample from which analytes are collected. In conventional microfluidic channels, ITP concentrates analytes from a sample volume of just a few hundred nanolitres. At low concentrations, the presence of target molecules in the volume sampled by ITP becomes probabilistic, and multiple parallel tests are required to avoid false negatives.
As a step towards large-volume sample processing, we developed a chip that enables ITP focusing of biological analytes from 50 μL sample volumes in less than 10 minutes. As all analytes from this volume are focused into an ITP interface comprising just half a nanolitre, the final concentration factor is more than 100,000, which is one hundred times more than ITP focusing in conventional microfluidic chips.
The key to processing larger sample volumes lies in combining a wide-channel region (with a large internal volume) and a tapering channel. However, this gradual geometry change and the concomitant Joule heating resulting from local differences in the current density lead to undesired dispersion of the sample plug, which is detrimental to the concentration of the focused analyte and ultimately compromises detection downstream. Fortunately, this undesired dispersion can be mitigated by using a ‘“recovery chamber” at the end of the chip. This makes the design highly scalable, and the capability to process even larger sample volumes on passively cooled chips is limited only by the Joule heating in the channels. Therefore, if the chips are actively cooled, sample volumes of hundreds of microlitres may be processed in this way.
We believe these results are of fundamental significance for the in vitro diagnostics and research community as they suggest the use of assays for improving the sensitivity and speed of molecular analysis, and for processing large volume of samples on devices with microfluidic elements.
References:
[1] F. Paratore, et al.:”Isotachophoresis-based surface immunoassay”, Analytical Chemistry, 2017, 89 (14), pp. 7373–7381.
[2] X.F. van Kooten, M. Truman-Rosentsvit, G. V. Kaigala, and M. Bercovici: “Focusing analytes from 50 μL into 500 pL: On-chip focusing from large sample volumes using isotachophoresis, Scientific Reports 7: 10467, 2017.
Please contact:
Moran Bercovici, Israel Institute of Technology
Govind V. Kaigala, IBM Research – Zurich, Switzerland











