Understanding the factors that influence human health and cause disease and using that understanding to develop treatments has always been the driving force behind medical research. In personalized medicine, the goal is to perform real-time analysis on a patient (using techniques such as magnetic resonance imaging) that can then be used to plan an individual course of treatment for that patient. Biomedical applications offer significant benefits both to the public and the scientific community, and consequently are emerging as challenging opportunities for innovation at the meeting point between medicine and computer science.
Frontiers in Medicine and HPC
In the world of high-performance computing, all signs point towards a future populated with multi-core heterogeneous processing technology. We are currently witnessing the emergence of multiple-core processors such as Intel Quad-Cores, graphics processing units and the Cell Broadband Engine® (Cell BE), which are bringing an unprecedented combination of low cost and low power to high-performance computing. A cooperative effort between the University of Basel and IBM is exploring the use of the Cell BE architecture in the individual biomedical planning of hyperthermia cancer therapy.
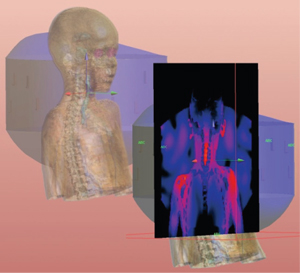
Hyperthermia is a promising treatment modality for various types of cancer. The technique involves heating the tumour with electromagnetic fields, generally using antenna arrays to focus the energy. In planning the therapy, the therapeutically optimal antenna parameters for the applicator are determined for each patient and the temperature distribution is predicted by solving the 3D Pennes bio-heat transfer equation. Although this can be a demanding task, a planning tool can greatly help clinical researchers to model and simulate the medical treatment. In this Cell BE project, we are addressing the practical concerns of fitting into a clinician's standard work-flow, and the question of how to use optimal algorithms and relevant HPC architectures to maximize application performance.
The Cell BE in a Nutshell
Cell BE is a heterogeneous multi-core processor that consists of one 64-bit power processing element (PPE) as the main processor and eight co-processors called synergistic processing elements (SPEs), which act as vector processors specialized for running computationally intensive applications. The latest chip generation of the Cell processor is now able to perform double-precision calculations with a peak near 200 GFlop/s. The SPEs are more specialized processors and consist of a synergistic processing unit (SPU) and a memory flow controller (MFC). An SPU is capable of performing SIMD (Single Instruction, Multiple Data) floating-point arithmetic and performs best when used in a streaming model. The MFC handles direct-memory access (DMA) transfers between the local SPE memory region of size 256 kB and the main memory of size 32 GB.
One of the radical changes in the Cell BE architecture is the elimination of the cache for the SPEs. The SPEs cannot access the main memory directly, so data required by the SPE must be explicitly transferred to and from the SPE local store by the use of asynchronous DMA transfers, which are handled by the MFC. The memory model, along with this specialization of tasks between SPEs and PPE, is a significant factor in the improvement in performance - approximately an order of magnitude - for key scientific applications and area-and-power efficiency.
Impact on Life Sciences
An order of magnitude improvement in performance over existing CPU technology is a disruptive change that can dramatically alter aspects of personalized medicine. A computational task that previously would have taken a year can now be completed in a few days; hour-long computations suddenly become interactive because they can be completed in seconds with the new Cell technology; and previously intractable real-time processing tasks now become tractable.

Since it is the temperature increase in hyperthermia cancer therapy that causes the increased cell death in tumours, it is of the utmost importance to determine the temperature distribution correctly and with high resolution. For this, the 3D Pennes bio-heat equation with temperature and time-dependent tissue parameters for the blood vessels can be solved interactively on the Cell architecture with 108 voxels. Validation of the Cell BE simulation is one of our future targets and will be performed with the Foundation for Research on Information Technologies in Society (IT'IS), Zurich, and the hyperthermia unit of the Erasmus Medical Center in Rotterdam.
Links:
http://fgb.informatik.unibas.ch/research/IBMFacultyAward/index.html
http://www-03.ibm.com/technology/cell
Please contact:
Olaf Schenk
University of Basel, Switzerland.
E-mail: olaf.schenk![]() unibas.ch
unibas.ch
Hema Reddy
IBM Corporation, Systems & Technology Group, Austin, US
E-mail: hemlata![]() us.ibm.com
us.ibm.com










